Sequential Changes after Radiofrequency Ablation and Cryoablation of Renal Neoplasms: Role of CT and MR Imaging
Satomi Kawamoto, MD, Sompol Permpongkosol, MD, PhD, David A. Bluemke, MD, PhD, Elliot K. Fishman, MD and Stephen B. Solomon, MD
Abstract
Radiofrequency (RF) ablation and cryoablation are increasingly being used as minimally invasive treatments for renal malignancies. Accurate assessment of ablated tumors at postprocedural imaging is essential for evaluating the adequacy of treatment and guiding further management. Renal tumors treated with RF ablation or cryoablation appear as low-attenuation regions at computed tomography (CT) and, relative to renal parenchyma, are generally hypointense at T2-weighted magnetic resonance (MR) imaging and iso- to hyperintense at T1-weighted imaging. The use of intravenous contrast material is essential for the evaluation of possible residual or recurrent tumor. At follow-up CT or MR imaging, successfully treated renal tumors appear as focal masses that demonstrate no evidence of contrast enhancement and that frequently decrease in size over time, whereas residual or recurrent tumor can be detected as abnormal foci of contrast enhancement. Follow-up surveillance imaging is warranted because long-term results for renal tumor ablation are not known, and evaluation for residual, recurrent, or metachronous tumor is essential.
Introduction
The increasing use of abdominal imaging has resulted in more frequent incidental detection of small asymptomatic renal tumors. Studies of patients with a diagnosis of renal cell carcinoma (RCC) conducted during various periods between 1989 and 1999 indicated that RCC was incidentally detected at cross-sectional imaging in 57%–61% of patients (1–4). Although radical nephrectomy has been considered the primary treatment for RCC, small, serendipitously found renal neoplasms are increasingly being treated with nephron-sparing procedures with partial nephrectomy (5).
Thermal ablative techniques that make use of radiofrequency (RF) heat ablation (6–17) or cryoablation (18–28) to destroy tumor cells have been developed to provide targeted therapy for renal tumors as an alternative to conventional surgical resection and are increasingly being used as minimally invasive treatments for renal malignancies, especially in patients who are poor candidates for surgical resection. Thermal ablative techniques can be performed with imaging-guided percutaneous approaches, intraoperative ultrasonography (US), or direct visualization during laparoscopic or open surgery. Percutaneous thermal ablative techniques that make use of RF ablation or cryoablation are the most recent developments in the management of small renal tumors (5).
RF ablation uses alternating electric current to produce ionic agitation and frictional heat in targeted tissue, resulting in coagulation necrosis and tumor desiccation (29). RF ablation of renal tumors is a relatively new technology whose use in humans with RCC was initially reported in 1997 (17). Initial results of RF ablation in the treatment of RCC are promising (7–16,30). In three recent studies with a mean follow-up of more than 2 years (excluding patients who died from unrelated causes or were lost to follow-up), successful local control was achieved for all small (<3-cm) exophytic tumors after one or two RF ablation sessions (7,11,12).
Cryoablation of renal tumors has been performed at open, laparoscopic, and retroperitoneoscopic surgery (19–25). However, the recent availability of small cryoprobes has increased the number of options available for percutaneous cryoablation (26–28). Intraoperative US can be used to monitor the cryoablated lesion at laparoscopic or open surgery. For percutaneous approaches, computed tomography (CT) or magnetic resonance (MR) imaging can be used to monitor ice balls during cryoablation that correlate closely with the ablation zones (31). An excellent correlation between the real-time US and pathologic measurements of cryoablated renal lesions has been reported in a dog model (32,33). Reported results of the use of cryoablation for local renal cancer control have also been encouraging. For example, four studies in which cryoablation was used with up to 3-year follow-up have shown no evidence of recurrence at contrast material–enhanced CT or MR imaging in 91%–95% of cryoablated renal tumors (20,23,26,28). However, larger trials with longer-term follow-up data are necessary to evaluate the efficacy of RF ablation and cryoablation of renal tumors.
Unlike with surgical resection, during thermal ablation therapy, the ablated renal tumor is not excised and is left in situ. Therefore, accurate assessment of ablated tumors and early postablation detection of residual or recurrent tumor is essential. The primary purpose of follow-up CT and MR imaging is to evaluate the adequacy of treatment and guide further management. Postprocedural CT and MR imaging are also used to evaluate for complications and for possible metastatic or metachronous tumors.
CT is often used for postablation follow-up because of its lower cost and wider availability; however, MR imaging may be superior in some situations, such as in patients who cannot receive iodinated contrast material because of renal insufficiency. For patients who underwent MR imaging–guided ablation, MR imaging may be better than CT for follow-up because intraprocedural findings can be directly compared with follow-up findings. Early imaging follow-up at 1–3 months after ablation is used to establish a baseline imaging appearance and to determine if the ablation procedure was complete or if there is residual tumor.
In this article, we discuss the appropriate time interval for follow-up CT or MR imaging of ablated renal tumors; describe CT and MR imaging protocols in this setting; and discuss and illustrate the spectrum of imaging appearances and sequential changes of renal tumors that have been successfully treated with RF ablation and cryoablation, as well as findings of residual or recurrent tumor.
Time Interval for Follow-up CT or MR Imaging
At our institution, patients usually undergo contrast-enhanced CT or MR imaging at approximately 3, 6, and 12 months after ablation and at 6–9-month intervals thereafter. CT is usually used as a first-line modality. At other institutions, early imaging follow-up has been performed within 1 month or less of ablation (6–11,15, 20,22,24,26). Most of the residual viable tumor was reported to be evident at 1–3 months after ablation (6).
The term residual tumor or incomplete treatment is used when there is focal enhancement in the tumor ablation zone at initial postprocedural imaging (usually performed 3 months after ablation at our institution). The term recurrent tumor is used when focal enhancement in the tumor ablation zone is not evident at initial postprocedural CT or MR imaging but does appear at subsequent follow-up imaging.
CT and MR Imaging Protocols
At our institution, follow-up CT is performed on a 16- or 64-section multi–detector row CT scanner. Three-phase helical CT scans are obtained, including an unenhanced scan and contrast-enhanced scans at 30 seconds (arterial phase) and 4–5 minutes (excretory phase) after the start of the intravenous bolus injection of 120 mL of non-ionic contrast material at a rate of 3 mL/sec. A nephrographic phase scan may also be obtained. A detector collimation of 16 × 0.75 mm or 64 × 0.6 mm, a 3-mm section thickness, and a 3-mm reconstruction interval were used for diagnostic reading. For volume rendering and multiplanar reformation, a 0.75-mm section thickness and a 0.5-mm reconstruction interval were used. Scanning parameters included 120 kV and 150–200 mAs.
In our opinion, arterial phase–excretory phase imaging is a reasonable alternative to nephrographic phase imaging in the detection of renal masses. Apaydin et al (34) reported no significant difference between nephrographic phase imaging performed 100 seconds after the initiation of contrast material injection and excretory phase imaging performed 300 seconds after the initiation of injection in the detection of renal masses 30 mm or less in diameter. Indeed, all lesions greater than 5 mm were detected at both nephrographic phase and excretory phase imaging. Furthermore, Yuh and Cohan (35) suggested that excretory phase imaging performed within 5 minutes of contrast material injection is an acceptable alternative to nephrographic phase imaging in assessing the kidneys for masses. We obtain arterial phase images as part of our protocol for CT angiography, which might be needed in patients who may require further surgical treatment. We also found that enhancing residual tumor may be better visualized on arterial phase images than on excretory phase images against the background of hypoattenuating ablated tumor (see Fig 5).
MR imaging is performed on a 1.5-T imager. Our MR imaging protocol is as follows: Axial un-enhanced T1-weighted images are obtained using two-dimensional spoiled gradient-recalled echo (repetition time msec/echo time msec = 200/2.8, flip angle = 80°) or three-dimensional fast spoiled gradient-recalled echo (4.1/1.6, flip angle = 15°) methods, and T2-weighted images are obtained using fast spin-echo (3,000/105, echo train length = 16) and single-shot fast spin-echo (∞/90) methods. Axial gadolinium-enhanced T1-weighted images are obtained at approximately 30 and 70 seconds after the injection of 0.1 mmol/kg of gadolinium chelate at a rate of 2 mL/ sec with either a two-dimensional or a three-dimensional technique. A breath-hold fat-suppressed MR imaging technique should be used. A coronal T1-weighted image is then obtained immediately after the axial images. A phased-array torso or body coil should be used for all acquisitions. Parallel imaging is not routinely performed, since signal-to-noise ratio may be compromised.
Postablation CT Features
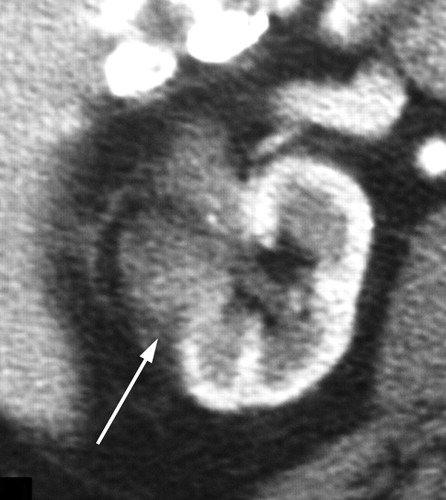
CT is widely used in the evaluation of renal tumors treated with RF ablation and cryoablation. Immediately after RF ablation, the ablated lesion is usually larger than the preablation tumor because a peripheral margin of normal renal tissue is also ablated. At follow-up CT, successfully ablated tumors are seen as unenhanced focal masses within the ablated lesion. Follow-up examinations show a decrease in the size of the ablated lesion. Unenhanced areas at CT performed after RF ablation are considered to correspond to the histopathologic areas of coagulation necrosis. Fat replacement at the interface between the ablated tumor and the normal kidney may be seen at follow-up examination due to shrinkage of the tumor (Fig 1) (36). Soft-tissue stranding in the perirenal or pararenal fat can be seen early after RF ablation. A curvilinear hyperattenuating area, or halo, is often seen within the perinephric fat surrounding the ablated lesion, a finding that roughly parallels but extends beyond the borders of the original renal tumor (Figs 1–3) (9).
Matsumoto et al (36) demonstrated evolution of the CT appearance of RF-ablated renal masses by analyzing 64 renal tumors with a mean follow-up of 13.7 months. After RF ablation, renal lesions within the kidney (ie, 75% of tumor volume within the boundary of the renal capsule) developed a low-attenuation, nonenhancing, wedge-shaped defect with fat infiltration between the ablated tissue and normal parenchyma, whereas exophytic tumors retained a configuration similar to that of the original tumor, with no contrast enhancement and only minimal shrinkage (36). The authors also reported that renal tumors treated with RF ablation tend to shrink slower than renal tumors treated with cryoablation (36).
Immediately after cryoablation, the ablated lesion appears at CT as a hypoattenuating area that is larger than the original tumor because the ice ball extends well beyond the visible margins of the targeted tumor (at least 5–7 mm, and typically about 1 cm) to achieve complete tumor cell death (20,21,37). At follow-up CT, successfully ablated lesions continue to manifest as hypoattenuating areas without focal contrast enhancement. In addition, the size of the cryoablated lesion decreases over time (Fig 4) (21). Renal tumors treated with cryoablation generally have a CT appearance similar to that of tumors treated with RF ablation, although cryoablated lesions may decrease in size faster than RF-ablated lesions (36). Soft-tissue stranding can be seen in the perirenal or pararenal fat early after cryoablation and decreases over time. As with RF ablation, a curvilinear band or halo may be seen within the perinephric fat surrounding the cryoablated lesion (Fig 5).
Intravenous contrast material is essential to evaluate for residual or recurrent tumor after RF ablation and cryoablation. Both unenhanced and contrast-enhanced CT are necessary to assess the degree of enhancement and to depict residual or recurrent tumor. Nodular enhancement after contrast material administration, or an increase in the size of the cryoablated lesion, should be considered to represent residual or recurrent tumor (Figs 3, 5, 6) (12). When tumor is present, the area of hypoattenuation at CT representing the ablated lesion may decrease in size; however, areas of nodular enhancement may continue to increase in size (Figs 5, 6). Contrast enhancement greater than 10 HU at CT is usually considered to represent persistent or recurrent tumor (8,36, 38). Incomplete treatment with the first RF ablation procedure is reportedly more common with large (3-cm), centrally located tumors (7,9,10).
Postablation MR Imaging Features
MR imaging also plays an important role in the evaluation of renal lesions after RF ablation and cryoablation, particularly in patients who are unable to receive iodinated contrast material for CT owing to renal insufficiency. To our knowledge, no systematic comparison of MR imaging with CT has been made to determine their relative accuracy at follow-up, and we currently consider the two imaging modalities to be diagnostically equivalent. MR imaging may be better suited for patients who have undergone MR imaging–guided ablation. In such patients, intraprocedural MR images can be directly compared with follow-up MR images, which helps to better understand and interpret findings (31).
RF ablation and cryoablation appear to cause similar effects and have similar manifestations at MR imaging (31). The area of coagulation necrosis resulting from RF ablation appears as an ovoid or round, generally hypointense region on T2-weighted images (39,40). This region is usually initially larger than the original tumor and decreases in size over time (Fig 7) (11,39). On T1-weighted images, the RF ablation zone is hyperintense relative to renal cortex in the majority of cases (Fig 7), although it may occasionally appear as a relatively iso- or hypointense region (39,40). Within the perinephric fat, a thin, hypointense border similar to that seen at post–RF ablation CT may appear, a finding that indicates the original extent of thermal injury (11).
Rukstalis et al (23) reported that 20 (87%) of 23 patients who underwent at least one 3-month follow-up MR imaging examination had complete resolution of the treated mass or a residual small parenchymal scar without enhancement, and that one patient had a persistent small, nonenhancing cystic lesion. In a report by Cestari et al (24) on 37 patients who underwent laparoscopic cryoablation, the mean diameter of solid renal masses was 25.7 mm; the mean diameter of the ablated lesion was 37.2 mm at 1 month after ablation, then decreased to 27.8 mm at 3 months and 19.7 mm at 6 months. At 24 months, the ablated lesions had nearly resolved, and only a renal scar was visible. In a study of 56 patients with renal tumors who underwent laparoscopic cryoablation, Gill et al (20) reported that the cryoablated tumor had decreased in size by an average of 75% 3 years after ablation relative to the cryoablated tumor on day 1, and that 38% of cryoablated tumors were not detectable at MR imaging at 3 years. The authors evaluated histopathologic specimens obtained from the cryoablated tumor beds 6 months after cryoablation and found fibrosis in 53% of cases, hemosiderin deposits in 28%, and coagulative necrosis in 12%. RCC was identified in two patients (20).
After RF ablation, rim enhancement of the ablated lesion may be seen on contrast-enhanced T1-weighted MR images, but this finding resolves gradually over time and is barely detectable after 3 months (39). After cryoablation, rim enhancement may occasionally be seen at early postprocedural MR imaging (20,23). This rim enhancement is believed to represent reactive changes in the area of sublethal injury and interstitial hemorrhage along the periphery of the ablated tumor (20) and typically disappears over time on subsequent MR images.
The MR imaging hallmark of successful renal tumor ablation is lack of tumor enhancement at gadolinium-enhanced imaging (20). Residual tumor following RF ablation has been reported to appear as hyperintense tissue on T2-weighted images and as enhancing tissue on contrast-enhanced T1-weighted images (39). Persistent nodular enhancement or an increase in the size of the ablated lesion on contrast-enhanced T1-weighted images should be taken to indicate locally persistent or recurrent cancer (20). Subtraction or quantitative assessment is used to evaluate contrast enhancement, since RF-ablated renal tumors often demonstrate high signal intensity on T1-weighted images.
Conclusions
Successfully ablated renal tumors appear as low-attenuation areas at CT and, relative to renal parenchyma, are generally hypointense at T2-weighted MR imaging and iso- to hyperintense at T1-weighted imaging.
Immediately after RF ablation and cryoablation, the ablated lesion appears larger than the original tumor because (a) a peripheral margin of normal renal tissue is also ablated with RF ablation, and (b) the ice ball created with cryoablation extends well beyond the visible margins of the targeted tumor.
At follow-up examination, ablated renal tumors are seen as focal masses without contrast enhancement that frequently decrease in size.
Intravenous contrast material is essential for the evaluation of possible residual or recurrent tumor, which any nodular foci of contrast enhancement within the ablated tumor should be considered to represent. Follow-up surveillance imaging is warranted because long-term results for renal tumor ablation are not known, and evaluation for recurrent or metachronous tumors is essential.
Figures
Abbreviations: RCC = renal cell carcinoma; RF = radiofrequency
References
1. LucianiLG, Cestari R, Tallarigo C. Incidental renal cell carcinoma: age and stage characterization and clinical implications—study of 1092 patients (1982–1997). Urology2000; 56(1): 58–62.
2. HsuRM, Chan DY, Siegelman SS. Small renal cell carcinomas: correlation of size with tumor stage, nuclear grade, and histologic subtype. AJR Am J Roentgenol2004; 182(3): 551–557.
3. JaysonM, Sanders H. Increased incidence of serendipitously discovered renal cell carcinoma. Urology1998; 51(2): 203–205.
4. LeeCT, Katz J, Fearn PA, Russo P. Mode of presentation of renal cell carcinoma provides prognostic information. Urol Oncol2002; 7(4): 135–140.
5. CohenHT, McGovern FJ. Renal-cell carcinoma. N Engl J Med2005; 353(23): 2477–2490.
6. GervaisDA, Arellano RS, McGovern FJ, McDougal WS, Mueller PR. Radiofrequency ablation of renal cell carcinoma. II. Lessons learned with ablation of 100 tumors. AJR Am J Roentgenol2005; 185(1): 72–80.
7. GervaisDA, McGovern FJ, Arellano RS, McDougal WS, Mueller PR. Radiofrequency ablation of renal cell carcinoma. I. Indications, results, and role in patient management over a 6-year period and ablation of 100 tumors. AJR Am J Roentgenol2005; 185(1): 64–71.
8. FarrellMA, Charboneau WJ, DiMarco DS, et al. Imaging-guided radiofrequency ablation of solid renal tumors. AJR Am J Roentgenol2003; 180(6): 1509–1513.
9. GervaisDA, McGovern FJ, Arellano RS, McDougal WS, Mueller PR. Renal cell carcinoma: clinical experience and technical success with radio-frequency ablation of 42 tumors. Radiology2003; 226(2): 417–424.
10. Mayo-SmithWW, Dupuy DE, Parikh PM, Pezzullo JA, Cronan JJ. Imaging-guided percutaneous radiofrequency ablation of solid renal masses: techniques and outcomes of 38 treatment sessions in 32 consecutive patients. AJR Am J Roentgenol2003; 180(6): 1503–1508.
11. LewinJS, Nour SG, Connell CF, et al. Phase II clinical trial of interactive MR imaging–guided interstitial radiofrequency thermal ablation of primary kidney tumors: initial experience. Radiology2004; 232(3): 835–845.
12. VarkarakisIM, Allaf ME, Inagaki T, et al. Percutaneous radio frequency ablation of renal masses: results at a 2-year mean followup. J Urol2005; 174(2): 456–460.
13. OganK, Jacomides L, Dolmatch BL, et al. Percutaneous radiofrequency ablation of renal tumors: technique, limitations, and morbidity. Urology2002; 60(6): 954–958.
14. PavlovichCP, Walther MM, Choyke PL, et al. Percutaneous radio frequency ablation of small renal tumors: initial results. J Urol2002; 167(1): 10–15.
15. VeltriA, De Fazio G, Malfitana V, et al. Percutaneous US-guided RF thermal ablation for malignant renal tumors: preliminary results in 13 patients. Eur Radiol2004; 14(12): 2303–2310.
16. HwangJJ, Walther MM, Pautler SE, et al. Radio frequency ablation of small renal tumors: intermediate results. J Urol2004; 171(5): 1814–1818.
17. ZlottaAR, Wildschutz T, Raviv G, et al. Radiofrequency interstitial tumor ablation (RITA) is a possible new modality for treatment of renal cancer: ex vivo and in vivo experience. J Endourol1997; 11(4): 251–258.
18. UchidaM, Imaide Y, Sugimoto K, Uehara H, Watanabe H. Percutaneous cryosurgery for renal tumours. Br J Urol1995; 75(2): 132–137.
19. GillIS, Novick AC, Soble JJ, et al. Laparoscopic renal cryoablation: initial clinical series. Urology1998; 52(4): 543–551.
20. GillIS, Remer EM, Hasan WA, et al. Renal cryoablation: outcome at 3 years. J Urol2005; 173(6): 1903–1907.
21. RodriguezR, Chan DY, Bishoff JT, et al. Renal ablative cryosurgery in selected patients with peripheral renal masses. Urology2000; 55(1): 25–30.
22. KhorsandiM, Foy RC, Chong W, Hoenig DM, Cohen JK, Rukstalis DB. Preliminary experience with cryoablation of renal lesions smaller than 4 centimeters. J Am Osteopath Assoc2002; 102(5): 277–281.
23. RukstalisDB, Khorsandi M, Garcia FU, Hoenig DM, Cohen JK. Clinical experience with open renal cryoablation. Urology2001; 57(1): 34–39.
24. CestariA, Guazzoni G, dell’Acqua V, et al. Laparoscopic cryoablation of solid renal masses: intermediate term followup. J Urol2004; 172(4 pt 1): 1267–1270.
25. BachmannA, Sulser T, Jayet C, et al. Retroperitoneoscopy-assisted cryoablation of renal tumors using multiple 1.5 mm ultrathin cryoprobes: a preliminary report. Eur Urol2005; 47(4): 474–479.
26. SilvermanSG, Tuncali K, vanSonnenberg E, et al. Renal tumors: MR imaging–guided percutaneous cryotherapy—initial experience in 23 patients. Radiology2005; 236(2): 716–724.
27. GuptaA, Allaf ME, Kavoussi LR, et al. Computerized tomography guided percutaneous renal cryoablation with the patient under conscious sedation: initial clinical experience. J Urol2006; 175(2): 447–453.
28. ShingletonWB, Sewell PE Jr. Percutaneous renal tumor cryoablation with magnetic resonance imaging guidance. J Urol2001; 165(3): 773–776.
29. GoldbergSN, Gazelle GS, Mueller PR. Thermal ablation therapy for focal malignancy: a unified approach to underlying principles, techniques, and diagnostic imaging guidance. AJR Am J Roentgenol2000; 174(2): 323–331.
30. McDougalWS, Gervais DA, McGovern FJ, Mueller PR. Long-term followup of patients with renal cell carcinoma treated with radio frequency ablation with curative intent. J Urol2005; 174(1): 61–63.
31. SilvermanSG, Tuncali K, Morrison PR. MR imaging-guided percutaneous tumor ablation. Acad Radiol2005; 12(9): 1100–1109.
32. OnikGM, Reyes G, Cohen JK, Porterfield B. Ultrasound characteristics of renal cryosurgery. Urology1993; 42(2): 212–215.
33. StephensonRA, King DK, Rohr LR. Renal cryoablation in a canine model. Urology1996; 47(5): 772–776.
34. ApaydinFD, Oezer C, Duce MN, et al. Comparison of reliabilities of the delayed images of helical renal CT in detecting small renal masses. J Clin Imaging2003; 27: 112–115.
CrossRef, Medline
35. YuhBI, Cohan RH. Different phases of renal enhancement: role in detecting and characterizing renal masses during helical CT. AJR1999; 173: 747–755.
36. MatsumotoED, Watumull L, Johnson DB, et al. The radiographic evolution of radio frequency ablated renal tumors. J Urol2004; 172(1): 45–48.
37. GillIS, Novick AC, Meraney AM, et al. Laparoscopic renal cryoablation in 32 patients. Urology2000; 56(5): 748–753.
38. GervaisDA, McGovern FJ, Wood BJ, Goldberg SN, McDougal WS, Mueller PR. Radio-frequency ablation of renal cell carcinoma: early clinical experience. Radiology2000; 217(3): 665–672.
39. MerkleEM, Nour SG, Lewin JS. MR imaging follow-up after percutaneous radiofrequency ablation of renal cell carcinoma: findings in 18 patients during first 6 months. Radiology2005; 235(3): 1065–1071.
40. BossA, Clasen S, Kuczyk M, et al. Magnetic resonance-guided percutaneous radiofrequency ablation of renal cell carcinomas: a pilot clinical study. Invest Radiol2005; 40(9): 583–590.
41. DelworthMG, Pisters LL, Fornage BD, von Eschenbach AC. Cryotherapy for renal cell carcinoma and angiomyolipoma. J Urol1996; 155(1): 252–255.